


ASICA is a research study to help understand how technology can be used to support people who have had skin cancer. The ASICA study is looking at how the NHS might use technology in the future to more effectively support people who have been treated for melanoma.
The incidence of melanoma has increased over four times since the late 1970s. Approximately 20% of patients treated for a melanoma experience a recurrence and 4-8% may develop a new melanoma. The risk of having a recurrence or developing a new melanoma is highest in the first five years after initial treatment. Current UK melanoma treatment guidelines recommend that all people treated for melanoma attend regular hospital follow-up appointments to help detect any recurrences or new melanomas that may have developed as soon as possible. Patients are also advised to regularly examine their own skin (total-skin-self-examinations) as some studies have found that most recurrences and new primaries are detected by patients between scheduled follow-up visits.

You may be eligible to take part in ASICA if:
If you would like any more information about ASICA, including information about taking part please contact the study office at the University of Aberdeen on 01224 438199 or email xrs-asica@abdn.ac.uk.

If you are eligible and would like to take part, your local hospital team will send you information about the ASICA study.
To take part you will be asked to sign a consent form and asked some questions about your health. After this you will be randomised to receive either the ASICA app in addition to your existing melanoma follow-ups, or continue with your existing melanoma follow-ups without the ASICA app.
We will follow your progress for 12 months and send you a confidential study questionnaire through the post or by your preferred method at 3, 6 and 12 months to ask about your health and any recurrence or new melanomas. The questionnaires can be returned to the study office in pre-paid envelopes.
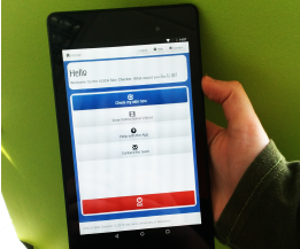

If you are randomised to receive ASICA, you will be invited to attend the hospital to have photographs of your skin taken in order to develop an individual skin map. The map will be then uploaded to a personalised hand-held ASICA computer tablet and only visible to you. You will receive training locally on how to undertake the total skin assessment, record it and send your results to the study specialist nurse for checking. In addition, you will receive an email or text message prompt to undertake monthly skin assessments in addition to your usual melanoma follow-ups. At the end of the year you will be asked to return the hand-held computer to the research team.
You will receive the same health care from your doctors whether or not you choose to participate in the study. You may not benefit personally from taking part but you will be directly helping us to plan the care of future patients with melanoma. The results of the study will help plan effective services offered by the NHS in the future.
We do not think that there are any possible disadvantages or risks to you in taking part in this study. Whichever group you are allocated to the follow-up care you are receiving for your melanoma will continue as standard of care at your local hospital.
You can decide at any time not to carry on with this study, but you should continue attending your usual melanoma follow-up appointments with your hospital doctor and/or GP as part of your standard care.
University of Aberdeen is the sponsor for this study based in the United Kingdom. We will be using information from you and your medical records in order to undertake this study and will act as the data controller for this study. This means that we are responsible for looking after your information and using it properly. University of Aberdeen will keep identifiable information about you at least 5 years after the study has finished.
Your rights to access, change or move your information are limited, as we need to manage your information in specific ways in order for the research to be reliable and accurate. If you withdraw from the study, we will keep the information about you that we have already obtained. To safeguard your rights, we will use the minimum personally-identifiable information possible.
You can find out more about how we use your information https://www.abdn.ac.uk/privacy.