


July 2020: Recruitment to PUrE has now restarted at some centres.
March 2020: In response to COVID-19, we have paused patient recruitment into this study. We are continuing to follow-up people who are already taking part. It may be that we are in touch with you by a different method than usual (for example we may telephone you to complete questionnaires, rather than send them by post). If you have any questions about the study, please get in touch by emailing pure@abdn.ac.uk.
If you are a patient and require medical attention or advice, please contact your GP or NHS-111/NHS-24 (by dialling 111).
You can also find information about COVID-19 from the NHS websites in England, Scotland, Wales and Northern Ireland.
There is also information about COVID-19 on the UK government website.
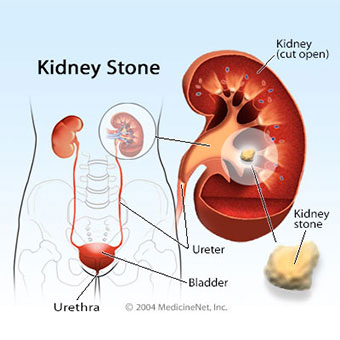
PUrE is a research study collecting information on the different ways of treating lower pole kidney stones. PUrE stands for the Percutaneous nephrolithotomy, flexible Ureterorenoscopy and Extracorporeal shockwave lithotripsy for lower pole kidney stone.
Kidney stone disease is very common (around 10% of adults will suffer in their lifetime) and mainly affects adults of working age (18 – 55 years). Over a quarter of patients with stones will require treatment and about half of all patients will go on to develop another stone.
Although some stones do not cause any problems, patients can develop serious symptoms including pain, infection, bloody urine and their kidneys can stop working as well as normal.
The lower pole (bottom) of the kidney is the most common location for stones in the kidney. These stones are more likely to require treatment, because they can be less likely to pass on their own.
You may be eligible to take part in PUrE if:
If you would like any more information about PUrE, including information about taking part please contact the study office at the University of Aberdeen on 01224 438156 or email pure@abdn.ac.uk.
If you are eligible and would like to take part in the PUrE study you will be recruited at your local hospital (please see ‘Where can I take part? to check if your local hospital is participating).
To take part you will be asked to sign a consent form and asked some questions about your health. You will also be asked to complete a study questionnaire.
After this if your stone is 10mm or smaller you will be randomised to receive either ESWL (ESWL.pdf) or FURS (Ureteroscopy_stone.pdf)
or if your stone is larger than 10 mm, but smaller than 25mm you will be randomised to receive either FURS (Ureteroscopy_stone.pdf) or PCNL (percutaneous_keyhole_removal_of_stones_from_kidney).
We will follow your progress for 18 months and we will send you a study questionnaire through the post at 3, 9, 12 and 18 months. We will also send you a prepaid envelope. The questionnaires are returned to the study office in a prepaid envelope.




University of Aberdeen/NHS Grampian is the sponsor for this study based in the United Kingdom. We will be using information from you and your medical records in order to undertake this study and will act as the data controller for this study. This means that we are responsible for looking after your information and using it properly. University of Aberdeen/NHS Grampian will keep identifiable information about you at least 10 years after the study has finished.
Your rights to access, change or move your information are limited, as we need to manage your information in specific ways in order for the research to be reliable and accurate. If you withdraw from the study, we will keep the information about you that we have already obtained. To safeguard your rights, we will use the minimum personally-identifiable information possible.
You can find out more about how we use your information https://www.abdn.ac.uk/privacy.